BABY
Finally, A Way to Prevent RSV Hospitalizations in Babies!
Here's what parents need to know about the new RSV shot for infants.

Written by
Happiest Baby Staff
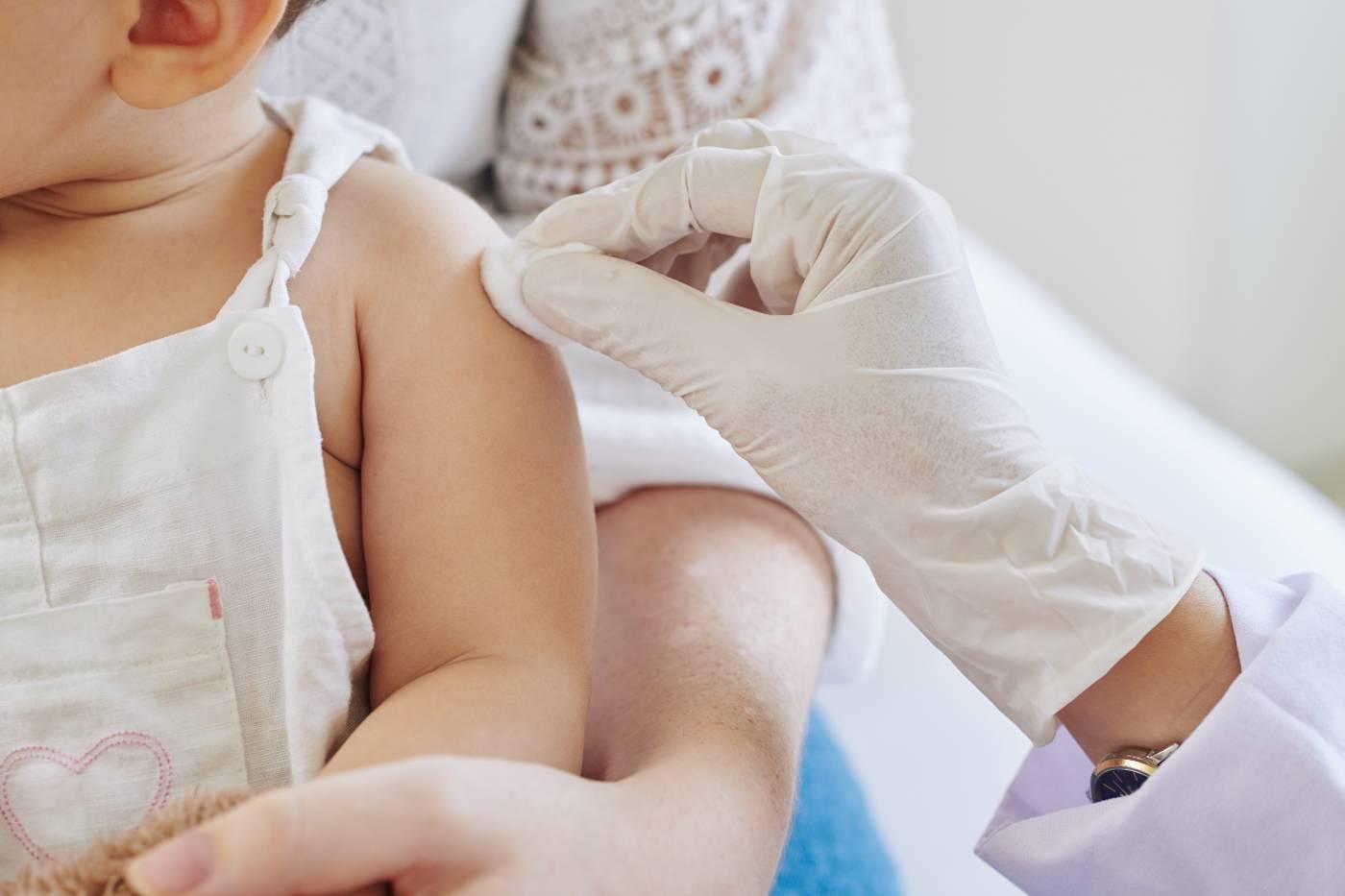
RSV (respiratory syncytial virus) is a very common—but sometimes scary—infection that puts up to 80,000 American children under the age of 5 in the hospital every year. While all little ones are vulnerable to RSV, premature infants, immunocompromised children, and babies under 6 months old are at an especially high risk for RSV complications like pneumonia or bronchiolitis, which can harm their teeny airways. Until recently, the most common ways to protect your child from RSV was to avoid close contact with sick people and diligently wash hands. But now, there’s a new way to shield your baby from RSV: the brand-new FDA-approved RSV shot for infants!
What is the RSV antibody shot?
In July 2023, the U.S. Food and Drug Administration (FDA) approved Beyfortus (nirsevimab) as the first monoclonal single-shot antibody to help prevent RSV in newborns, babies, and toddlers. The same RSV shot is already approved in Canada, Europe, and the UK. (Learn more about RSV.)
What is the difference between a vaccine and an antibody?
Most vaccines contain a weakened form of the pathogen that directs your immune system to develop antibodies, but the RSV antibody shot is different! Beyfortus sends protective antibodies directly to the bloodstream to boost immunity. Plus, while traditional vaccines can take a few weeks to spur an immune response, a monoclonal antibody shot works almost instantly.
How effective is the RSV shot?
Three separate clinical trials have shown that the RSV antibody reduces the risk of RSV infection between 70% and 75% among infants and children aged 2 and younger. And, according to the CDC, the RSV shot reduces the risk of severe RSV by 80%. Unlike a lot of vaccines that offer long-term protection, a monoclonal antibody treatment, like the RSV shot, is only effective for a few months. A single dose of Beyfortus has been shown to protect against RSV for about five months, which is usually the length of RSV season.
Who is the new RSV shot for?
The Centers for Disease Control and Prevention (CDC) recommends RSV immunization for for all newborns and babies who are entering their first RSV season. (RSV season starts in the fall, peaks in the winter, and drags on to early spring.) Additionally, the CDC recommends the RSV shot for children between 8 and 19 months who are extra-vulnerable to severe RSV infection—such as children with long-term breathing and lung problems or those with congenital heart disease—who are entering their second RSV season.
Is there an RSV vaccine for adults?
Yes! The FDA has approved two RSV vaccines to protect adults 60 and older from the virus. This is very important since between 60,000 and 160,000 older Americans are hospitalized with RSV every year—and up to 10,000 die. Plus, when Grandma or Grandpa get their RSV shot, they’re not only helping to keep themselves safe, they’re helping to prevent their grandbabies from being exposed to RSV, too. The FDA has also approved an RSV vaccine for pregnant folks that’s meant to protect infants from the virus for about the first 6 months of the baby’s life.
Should I get the RSV vaccine during pregnancy?
The American College of Obstetricians and Gynecologists (ACOG) recommends the Pfizer RSV vaccine for folks who are between 32 and 36 weeks pregnant from September to January (basically for parents expecting a fall or winter baby). That's because the RSV vaccine for pregnant people makes antibodies that you can pass along to your growing fetus, which gives them protection in their first months of life.
If you qualify for the RSV vaccine while pregnant, you will likely be given two options: to get vaccinated during pregnancy or to get your baby the RSV shot after they're born. Your OB/GYN can provide additional guidance, but some things to consider include:
- If you get the vaccine during pregnancy, your baby will likely be protected as soon as they're born
- Getting the vaccine yourself means that there's one less shot your baby will have to get
- The antibody shot given after birth may offer longer protection
- The antibody shot for babies has been hard to come by, so you may have better luck finding the adult vaccine
Is the new RSV antibody shot safe?
All research points to yes! Most common side effects are mild, like a possible rash and injection site reactions. That said, doctors need to exercise caution before administering Beyfortus to little ones with bleeding disorders like hemophilia. And anyone with a history of serious reactions to any of Beyfortus’ active ingredients should skip the shot. Don’t hesitate to talk to your pediatrician if you have any questions!
When will the RSV shot be available?
While the RSV antibody shot is available, it has been in short supply. Because it's so limited, pediatricians have been prioritizing the babies and toddlers who are at greatest risk from RSV, including:
- Babies between 6 and 7 months with preexisting medical conditions that put them at high risk
- Babies weighing less than 11 pounds
- Babies under 6 months who weigh more than 11 pounds
- Babies under 8 months who identify as American Indian and Alaska Native
- Babies between 8 to 19 months who identify as American Indian or Alaska Native and live in a remote region
Bottom Line on the RSV Shot
For most toddlers and older kids, RSV is not a big deal. The standard runny nose, cough, and fever is nothing that some rest, fluids, and TLC can’t take care of. However, RSV is no joke for many babies, especially premature infants, babies under 6 months, and those with certain medical conditions. For vulnerable kids, RSV can get worse—and dangerous—very fast, causing serious lung infections, which can hinder breathing and cause hospitalization. For others, RSV irritates the lungs so much that their chesty cough can linger for many months.
Now that there’s a very effective and safe way to shield your baby from this type of sickness, I strongly urge parents to get their little ones protected!
You May Also Be Interested In…
- Your Cold, Flu, and RSV Toolkit for Babies
- Vaccine Guide and Baby Immunization Schedule
- The Danger of Antibiotic Overuse—and What Parents Can Do
- How to Help Toddlers Deal With Shots
Disclaimer: The information on our site is NOT medical advice for any specific person or condition. It is only meant as general information. If you have any medical questions and concerns about your child or yourself, please contact your health provider.
SHARE THIS ARTICLE












